Methodology: A retrospective study analyzing thyroid FNAs collected over a 20-month period was designed to determine if use of the Afirma GEC has impacted clinical practice at UCLA. The study analyzed 174 patient with Afirma GEC results compared to previously published data from their institution before the implementation of the Afirma GEC.
Findings: Among the 174 patients with indeterminate cytology, Afirma GEC identified 80 as benign (46%). Only 5 of the 80 patients (6.3%) underwent surgery and all were benign on histopathology. When compared to previously published data it was found that implementation of Afirma GEC reduced the overall surgical rate on cytology indeterminate patients while increasing the overall rate of cancer found after surgical resection (Figure 1).
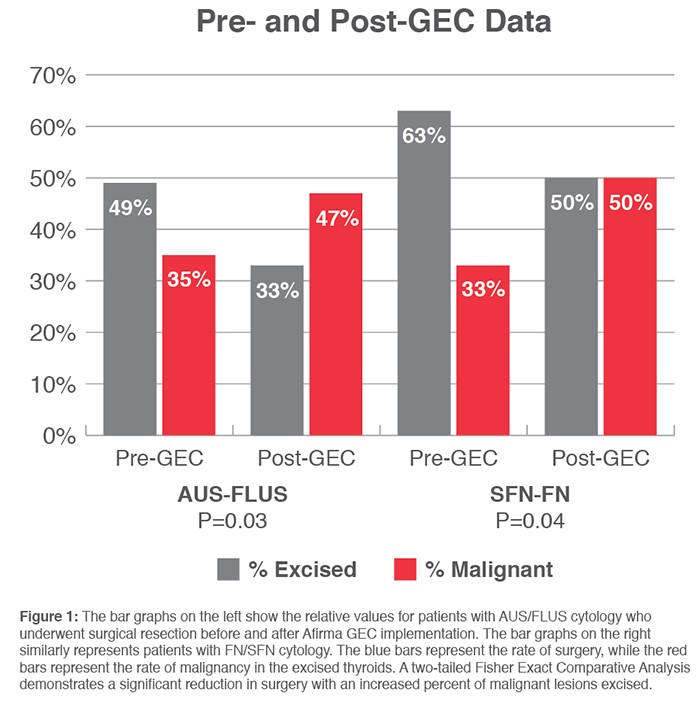
Author’s Conclusions:
“The question remains whether Afirma GEC testing has refined the indeterminate thyroid category in cytology. The answer in our opinion is a qualified ‘yes’ because it excludes approximately 40% to 50% of the benign cases from surgery and results in a relatively higher percentage of malignant lesions in the surgical outcome.”